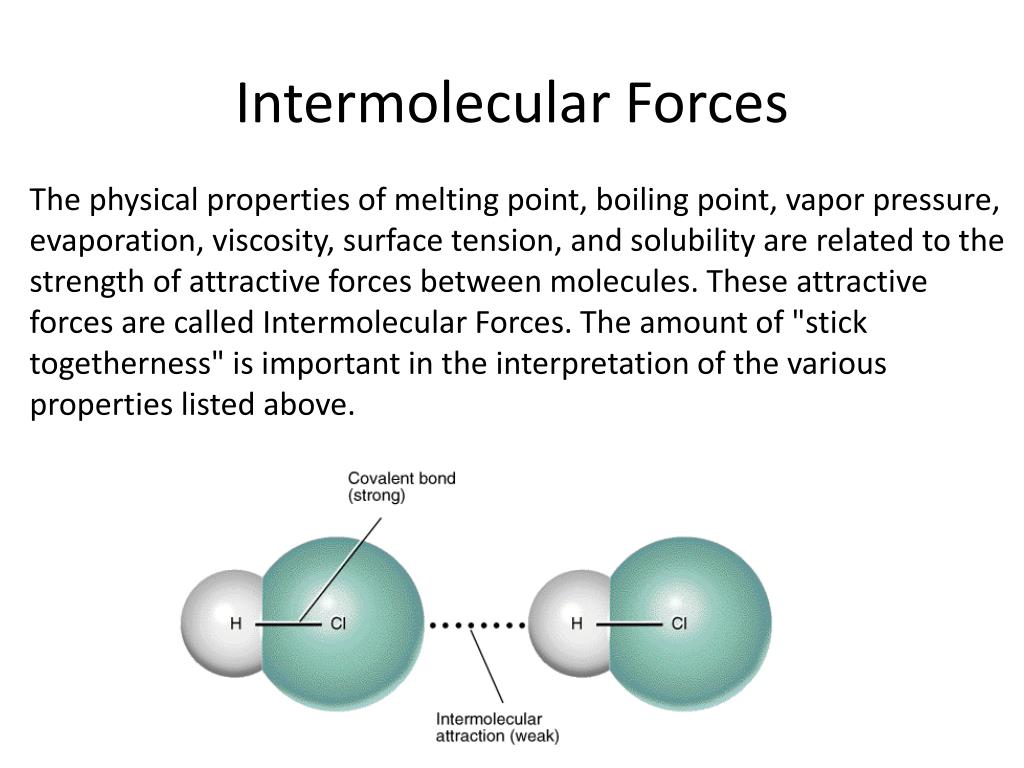
Does High Surface Tension Mean Strong Intermolecular Forces . The core principle is that the stronger the imfs in the sample of molecules, the more. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. having strong intermolecular forces means that the attraction between the molecules is strong. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface tension. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. as a result, molecules are held together by strong hydrogen bonding. As a result of this high surface tension, the. Since water has a high. Therefore, if a substance has stronger imfs, it is harder to pull its. Energy is required to break these intermolecular bonds. The dashed line shows the shape the. surface tension exerts a downward force as it flattens the mercury, suppressing it in the tube.
from www.slideserve.com
Therefore, if a substance has stronger imfs, it is harder to pull its. Since water has a high. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. As a result of this high surface tension, the. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface tension. as a result, molecules are held together by strong hydrogen bonding. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. The core principle is that the stronger the imfs in the sample of molecules, the more. The dashed line shows the shape the. Energy is required to break these intermolecular bonds.
PPT Intermolecular Forces PowerPoint Presentation, free download ID
Does High Surface Tension Mean Strong Intermolecular Forces having strong intermolecular forces means that the attraction between the molecules is strong. having strong intermolecular forces means that the attraction between the molecules is strong. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. The dashed line shows the shape the. As a result of this high surface tension, the. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface tension. as a result, molecules are held together by strong hydrogen bonding. Therefore, if a substance has stronger imfs, it is harder to pull its. among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. Energy is required to break these intermolecular bonds. surface tension exerts a downward force as it flattens the mercury, suppressing it in the tube. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. Since water has a high. The core principle is that the stronger the imfs in the sample of molecules, the more.
From slideplayer.com
Intermolecular Forces and ppt download Does High Surface Tension Mean Strong Intermolecular Forces The dashed line shows the shape the. among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. Energy is required to break these intermolecular bonds. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. surface tension exerts a downward force as it. Does High Surface Tension Mean Strong Intermolecular Forces.
From slideplayer.com
Intermolecular forces ppt download Does High Surface Tension Mean Strong Intermolecular Forces The dashed line shows the shape the. as a result, molecules are held together by strong hydrogen bonding. As a result of this high surface tension, the. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Intermolecular forces Generalizing properties PowerPoint Does High Surface Tension Mean Strong Intermolecular Forces The core principle is that the stronger the imfs in the sample of molecules, the more. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. having strong intermolecular forces means that the attraction between the molecules is strong. liquids that have strong intermolecular forces, like the hydrogen bonding. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Intermolecular Forces PowerPoint Presentation, free download ID Does High Surface Tension Mean Strong Intermolecular Forces as a result, molecules are held together by strong hydrogen bonding. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. As a result of this high surface tension, the. having strong intermolecular forces means that the attraction between the molecules is strong. Therefore, if a substance has stronger imfs, it. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.scienceabc.com
Surface Tension Definition, Explanation, Examples And Significance Does High Surface Tension Mean Strong Intermolecular Forces having strong intermolecular forces means that the attraction between the molecules is strong. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. The core principle is that the stronger the imfs in the sample of molecules, the more. liquids that have strong intermolecular forces, like the hydrogen bonding in water,. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Intermolecular Forces PowerPoint Presentation, free download ID Does High Surface Tension Mean Strong Intermolecular Forces surface tension exerts a downward force as it flattens the mercury, suppressing it in the tube. The dashed line shows the shape the. Therefore, if a substance has stronger imfs, it is harder to pull its. Energy is required to break these intermolecular bonds. as a result, molecules are held together by strong hydrogen bonding. liquids that. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.youtube.com
Surface tension and capillary action Intermolecular Forces meriSTEM Does High Surface Tension Mean Strong Intermolecular Forces The core principle is that the stronger the imfs in the sample of molecules, the more. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. having strong intermolecular forces means that the attraction between the molecules is strong. liquids that have strong intermolecular forces, like the hydrogen bonding in water,. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.youtube.com
Intermolecular Forces Trends Melting & Boiling Point, Viscosity Does High Surface Tension Mean Strong Intermolecular Forces Therefore, if a substance has stronger imfs, it is harder to pull its. The dashed line shows the shape the. as a result, molecules are held together by strong hydrogen bonding. having strong intermolecular forces means that the attraction between the molecules is strong. Since water has a high. for example, water, with its strong intermolecular hydrogen. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Intermolecular Forces and Liquids and Solids PowerPoint Does High Surface Tension Mean Strong Intermolecular Forces The dashed line shows the shape the. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. having strong intermolecular forces means that the attraction between the molecules is strong. Energy is required to break these intermolecular bonds. Therefore, if a substance has stronger imfs, it is harder to pull. Does High Surface Tension Mean Strong Intermolecular Forces.
From ceketiqv.blob.core.windows.net
Liquid Surface Tension Is Due To Which Molecular Force at Nunez blog Does High Surface Tension Mean Strong Intermolecular Forces Therefore, if a substance has stronger imfs, it is harder to pull its. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. Since water has a high. As a result of this high. Does High Surface Tension Mean Strong Intermolecular Forces.
From byjus.com
Explain the surface tension phenomenon with examples. Does High Surface Tension Mean Strong Intermolecular Forces liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface tension. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. as a result, molecules are held together by strong hydrogen bonding. The dashed line shows the shape the. The core principle is that the. Does High Surface Tension Mean Strong Intermolecular Forces.
From fity.club
Intermolecular And Surface Forces Does High Surface Tension Mean Strong Intermolecular Forces Since water has a high. Energy is required to break these intermolecular bonds. as a result, molecules are held together by strong hydrogen bonding. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. surface tension exerts a downward force as it flattens the mercury, suppressing it in the. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Chapter 11 Liquids and Intermolecular Forces PowerPoint Does High Surface Tension Mean Strong Intermolecular Forces among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface tension. for example, water, with its strong intermolecular hydrogen bonding, has one of the highest surface tension values of. Energy is required to. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Intermolecular Forces PowerPoint Presentation, free download Does High Surface Tension Mean Strong Intermolecular Forces As a result of this high surface tension, the. Since water has a high. The core principle is that the stronger the imfs in the sample of molecules, the more. liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface. having strong intermolecular forces means that the attraction between the molecules is. Does High Surface Tension Mean Strong Intermolecular Forces.
From isaacscienceblog.com
Intermolecular forces Isaac's science blog Does High Surface Tension Mean Strong Intermolecular Forces The core principle is that the stronger the imfs in the sample of molecules, the more. As a result of this high surface tension, the. surface tension exerts a downward force as it flattens the mercury, suppressing it in the tube. having strong intermolecular forces means that the attraction between the molecules is strong. liquids that have. Does High Surface Tension Mean Strong Intermolecular Forces.
From slideplayer.com
Intermolecular Forces and ppt download Does High Surface Tension Mean Strong Intermolecular Forces liquids that have strong intermolecular forces, like the hydrogen bonding in water, exhibit the greatest surface tension. Since water has a high. The core principle is that the stronger the imfs in the sample of molecules, the more. The dashed line shows the shape the. having strong intermolecular forces means that the attraction between the molecules is strong.. Does High Surface Tension Mean Strong Intermolecular Forces.
From chem.libretexts.org
11.4 Intermolecular Forces in Action Surface Tension, Viscosity, and Does High Surface Tension Mean Strong Intermolecular Forces Since water has a high. among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. surface tension exerts a downward force as it flattens the mercury, suppressing it in the tube. as a result, molecules are held together by strong hydrogen bonding. liquids that have strong intermolecular forces,. Does High Surface Tension Mean Strong Intermolecular Forces.
From www.slideserve.com
PPT Implication of Intermolecular Forces PowerPoint Presentation Does High Surface Tension Mean Strong Intermolecular Forces Therefore, if a substance has stronger imfs, it is harder to pull its. surface tension exerts a downward force as it flattens the mercury, suppressing it in the tube. Energy is required to break these intermolecular bonds. among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. having strong. Does High Surface Tension Mean Strong Intermolecular Forces.